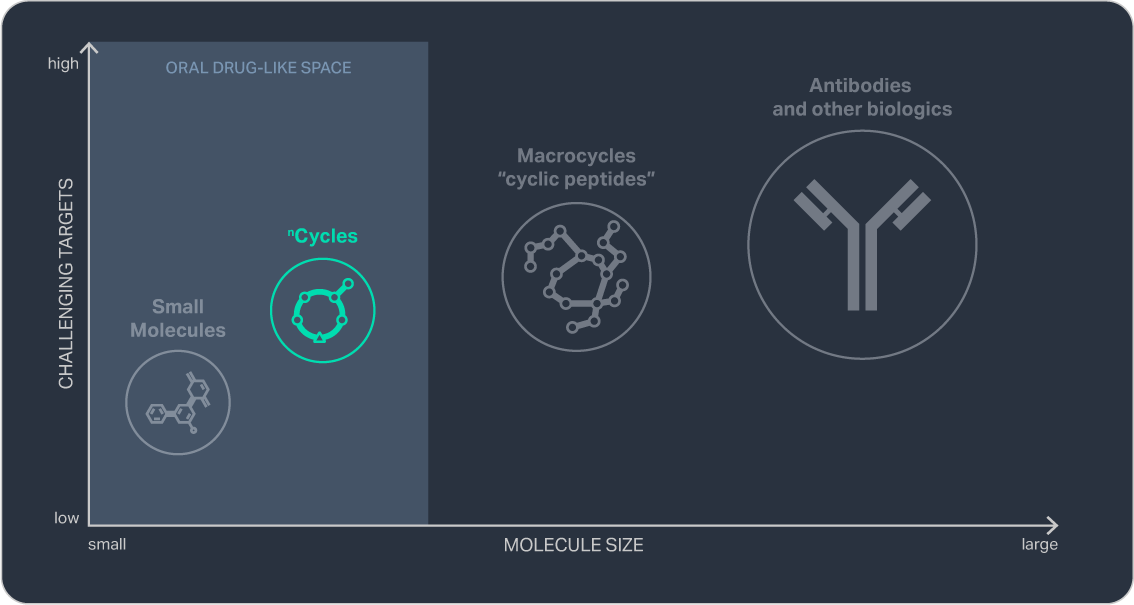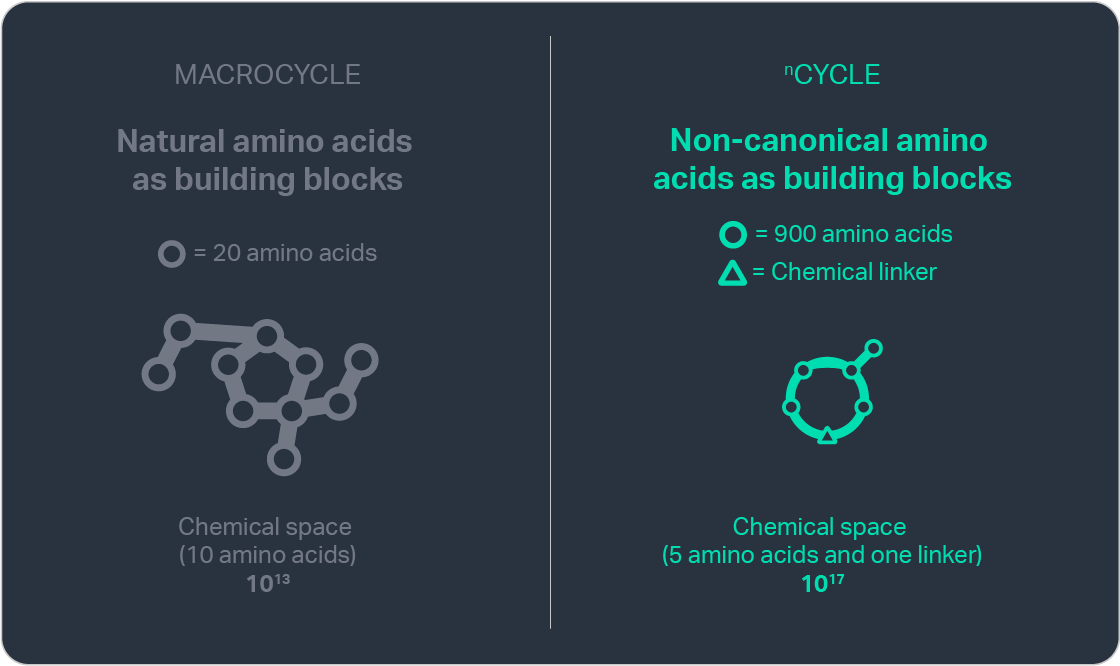
To enhance the therapeutic potential of macrocycles, we have designed nCycles — a new molecular class engineered for increased oral bioavailability by combining reduced size with expanded chemical diversity, including amino acids beyond those found in nature. nCycles can be manufactured much like small-molecule APIs, enabling bulk production through highly scalable processes with fast lead times and low cost of goods.
nCycles (500-1000 daltons) are typically less than half the size of first-generation macrocycles (1,500-2,500 daltons). This more compact size gives nCycles an edge in membrane permeability and oral bioavailability.
By extending beyond nature’s amino acid repertoire, our chemistry accesses a design space of up to 1030 possible structures, a number greater than the sum of stars in the observable universe. This breadth allows us to discover molecules that are finely tuned to their biological targets while exhibiting optimized drug-like characteristics.
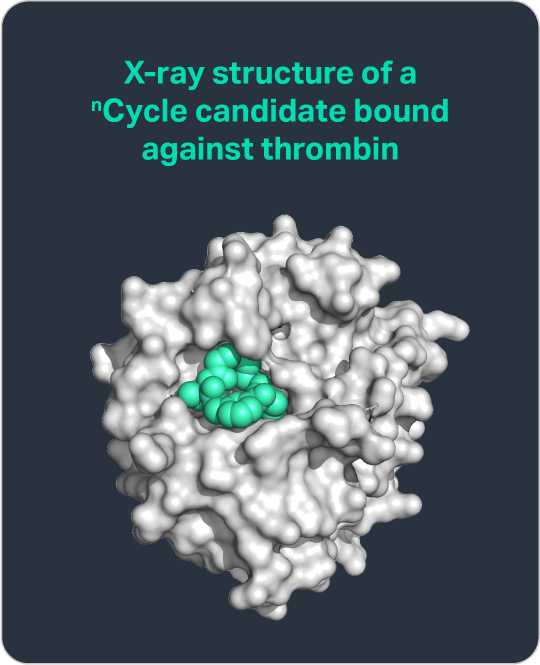
The potential of nCycles has been validated through an exceptional proof-of-concept demonstrated by our scientific founder Christian Heinis. Using a high throughput approach that we have since expanded upon to create our nGen discovery platform, Dr. Heinis identified a thrombin inhibitor with 18% oral bioavailability in rats—a marked improvement over the industry standard of 1-2%.
Published in Nature Chemical Biology, this work highlights the broader potential of nCycles to enable orally available therapeutics across a wide range of protein targets and disease indications.
Published in Nature Chemical Biology, this work highlights the broader potential of nCycles to enable orally available therapeutics across a wide range of protein targets and disease indications.